Biomolecules Class 12 Notes Chemistry Chapter 14 - CBSE
Chapter:14
What are Biomolecules ?
To download the complete Syllabus (PDF File), Please fill & submit the form below.
Carbohydrates
Carbohydrates are defined as optically active polyhydroxy aldehydes or polyhydroxy ketones or the compounds which can be hydrolysed to them.
Classification of Carbohydrates
| Monosaccharides | Oligosaccharides | Polysaccharides |
|
|
|
| Cannot be hydrolysed into simpler compounds | E.g, Sucrose, Lactose, Maltose | E.g. Cellulose, starch |
| E.g., Glucose, mannose |
Preparation Of Glucose
From Sucrose :
$$\\\underset{\mathrm{Sucrose}}{C_{12}H_{22}O_{11} + H2_O}\xrightarrow[]{H+}\underset{\mathrm{Glucose}}{C_6H_{12}O_6}\space\underset{\mathrm{Fructose}}{C_6H_{12}O_6}$$
From Starch :
$$\\\underset{\mathrm{Starch or cellulose}}{(C_6H_{10}O_5)_n}+nH_2O\xrightarrow[393K: 2.3 atm]{H+}\underset{\mathrm{Glucose}}{nC_6H_{12}O_6}$$
Cyclic Structure Of Glucose And Fructose

Disaccharides
A disaccharide (also called a double sugar or biose) is the sugar formed when two monosaccharides are joined by glycosidic linkage. The linkage between two monosaccharide units through oxygen atom is called glycosidic linkage.
Examples Of Disacchar Ides
Sucrose (glucose and fructose)

Lactose (galactose and glucose)

Maltose (glucose and glucose)

Polysaccharides
Polysaccharides contain a large number of monosaccharide units which are joined together by glycosidic linkages. These are the most common carbohydrates found in nature which mainly act as the food storage.
Examples of Polysaccharides
Starch : Made up of water-soluble content amylose and water insoluble content amylopectin.


Cellulose : It is the most important constituent of cell wall in plants.

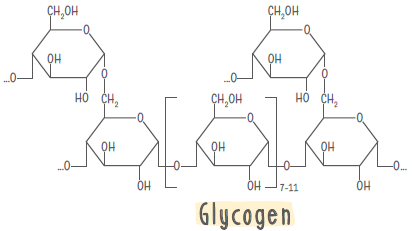
Glycogen : It is also known as animal starch and is stored in animal body as glycogen.
Proteins
They are the complex organic substances which are the bases of protoplasm and are found all living organism. They are polymers of amino acids.
Amino Acids: These are amino substituted carboxylic acids. α-amino acids are the building blocks of peptides and proteins.

Zwitter ion structure:

In acidic solution amino acids exists as cations and migrate towards cathode in an electric field whereas in basic solution they exists as anions and migrate towards anode. At the intermediate pH, however, they exists as zwitter ion (a dipolar ion) and do not migrate towards either electrode. The pH is known as isoelectric
point of ∝-amino acid.
Classification of Proteins
Based on their molecular shape, proteins are classified as:
| Fibrous Proteins | Globular Proteins |
| These proteins have fibre-like structure and are formed when the polypeptide chains run parallel and are held together by hydrogen and disulphide bonds. Such proteins are generally insoluble in water. Some common examples are keratin (present in hair, wool, silk) and myosin (present in muscles), etc. | These proteins are formed when the chains of polypeptides coil around to give a spherical shape. These are usually soluble in water. Insulin and albumins are the common examples of globular proteins. |
Classification Of Amino Acids Based On Their Synthesis In Our Body
All proteins are polymers of ∝-amino acids. Proteins are the most abundant biomolecules of the living system.
They are required for growth and maintenance of body. The amino acids are the compounds containing at least one amino (–NH2) and one carboxyl (–COOH) functional group. Based on the synthesis in our body, amino acids are classified as: Essential and Non-essential Amino acids.
- Essential Amino Acids: Certain amino acids are required for proper health and growth in human beings. But the human body is unable to synthesis them. These must be supplied to body through food and are called essential amino acids. E.g. Histidine, Isoleucine, Leucine, Lysine, Methionin, Phenylalanine, Threonine,
Tryptophan, Valine. - Non-essential Amino Acids: There are 20 different amino acids, each with a different - R group, commonly found, in the proteins of living things. The human body can synthesis 10 out of the 20 amino acids found in the proteins. These are called non-essential amino acids. E.g. Alanine, Arginine, Asparagine, Aspartic acid,
Cysteine, Glutamic acid, Glutamine, Glycine, Proline, Selenocysteine, Serine, Tyrosine.
Structure of Proteins
| Primary protein structure is sequence of a chain of amino acids. | Secondary protein structure occurs when the sequence of amino acids are linked by hydrogen bonds. | Tertiary protein structure occurs when certain attractions are present between alpha helices and pleated sheets. | Quaternary protein structure is a protein consisting of more than one amino acid chain. |
Denaturation Of Proteins
Protein found in a biological system with a definite configuration, unique three-dimensional structure and biological activity is called a native protein. When a native protein is subjected to physical change or chemical change like change in temperature or change in pH, the hydrogen bonds are disturbed. Due to this, globules unfold and helix get uncoiled and protein loses its biological activity. This is called denaturation of protein. During denaturation secondary and tertiary structures are destroyed but primary structure remains intact.
Enzymes
Enzymes are naturally occurring simple or conjugated proteins that catalyse biological reactions. Almost all the enzymes are globular proteins and they are very specific for a particular reaction and particular substrate. They are generally named after the compound upon which they work. For example, the enzyme that catalyses hydrolysis of maltose into glucose is named as maltase.
$$\\\underset{\mathrm{Starch or cellulose}}{C_{12}H_{22}O_{11}(aq)}+ H_2O(I)\xrightarrow[]{Maltase}\underset{\mathrm{Glucose}}{2C_6H_{12}O_6(aq)}$$
Vitamins
Vitamins are substances necessary to maintain normal health, growth and nutrition. They are generally required in the diet in small amounts to perform specific biological functions for normal maintenance and health of the organism.
Classification of Vitamins
| Fat-soluble vitamins | Water-soluble vitamins |
| Vitamins which are insoluble in water but are soluble in fat are called as fat-soluble vitamins. A, D, E and K are fat soluble vitamins and they are stored in liver and adipose tissues. | Vitamins which are insoluble in fat but soluble in water are called as water soluble vitamins. Vitamins B and C are water soluble. |
Nucleic Acids
Nucleic acids are long chain polymers that are present in high concentration in the nucleic of cells. They play vital role in the cell replication and protein synthesis. They are of two types :
(a) DNA (DeoxyriboNucleic Acid)
(b) RNA (RiboNucleic Acid)
The nitrogen containing bases found in nucleotides are derivatives of either purine or pyrimidine :
Bases in Nucleic Acids
Pyrimidine

The base thyrmine is found only in nucleotides of DNA and uracil is found in RNA. The second component of the nucleotides is the sugar molecule. The sugar present in RNA is D-ribose and DNA is D-2-deoxyribose.