NCERT Solutions for Class 12 Chemistry Chapter 11 - Alcohols, Phenols and Ethers
NCERT Solutions for Class 12 Chemistry Chapter 11 Free PDF Download
Please Click on Free PDF Download link to Download the NCERT Solutions for Class 12 Chemistry Chapter 11 Alcohols, Phenols and Ethers
To download the complete Syllabus (PDF File), Please fill & submit the form below.
1. Write IUPAC names of the following compounds:
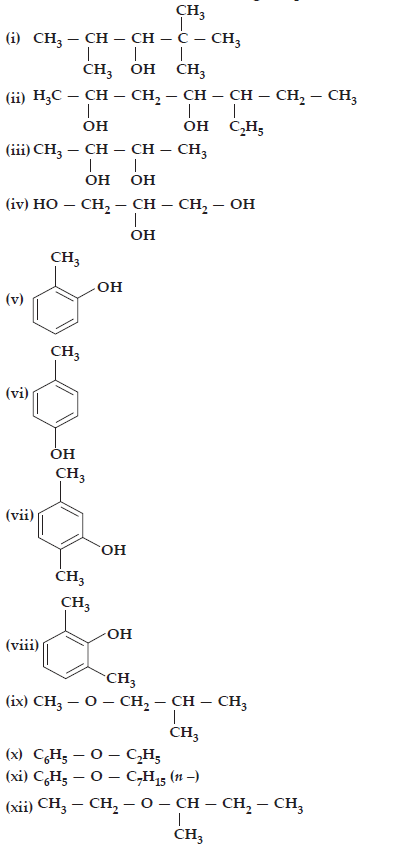
Ans. (i) 2,2,4-trimethylpentan-3-ol
(ii) 5-ethylheptane-2,4-diol
(iii) Butane-2,3-diol
(iv) Propane-1,2,3-triol
(v) 2-methylphenol
(vi) 4-methylphenol
(vii) 2,5-dimethylphenol
(viii) 2,6-dimethylphenol
(ix) 1-methoxy-2-methylpropane
(x) Ethoxybenzene
(xi) 1-phenoxyheptane
(xii) 2-ethoxybutane
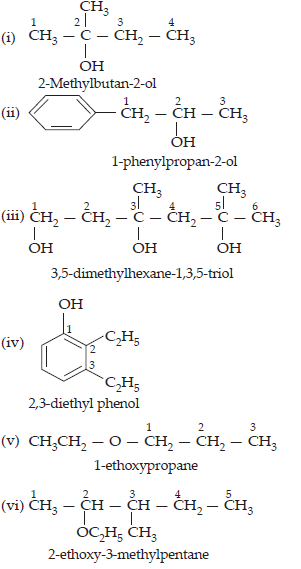
2. Write structures of the compounds whose IUPAC names are as follows:
(i) 2-methylbutan-2-ol
(ii) 1-phenylpropan-2-ol
(iii) 3,5-dimethylhexane-1,3,5-triol
(iv) 2,3-diethylphenol
(v) 1-ethoxypropane
(vi) 2-ethoxy-3-methylpentane
(vii) Cyclohexylmethanol
(viii) 3-cyclohexylpentan-3-ol
(ix) Cyclopent-3-en-1-ol
(x) 4-chloro-3-ethylbutan-1-ol
Ans.

3. (i) Draw the structures of all the isomeric alcohols of molecular formula C5H12O and give their IUPAC names.
(ii) Classify the isomers of alcohols in question (a) primary, secondary and tertiary alcohols.'
Ans. (i) The molecular formula C5H12O represents eight isomeric alkanols. These are:

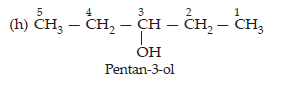
(ii) Primary: (a), (b), (c) and (d); Secondary: (e), (f) and (h); Tertiary: (g)
4. Explain why propanol has higher boiling point than that of the hydrocarbon, butane?
Ans. The molecules of butane are held together by weak van der Waal’s forces of attraction while those of propanol are held together by stronger intermolecular hydrogen bonding.
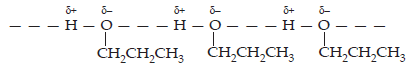
Therefore, the boiling point of propanal is much higher than that of butane.
5. Alcohols are comparatively more soluble in water than hydrocarbons of comparable molecular masses. Explain the fact.
Ans. Water and alcohols both are polar in nature. When an alcohol is dissolved in water, it forms H-bonds with water molecule by breaking the H-bond already existing between water molecules.
Hydrocarbons are non-polar in nature and do not forms H-bonds with w ater molecules. Therefore, alcohols are readily soluble in water whereas hydrocarbons are not.
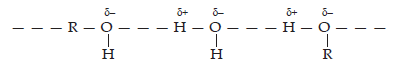
Hydrogen bonding among alcohol and water.
6. What is meant by hydroboration-oxidation reaction? Illustrate it with an example.
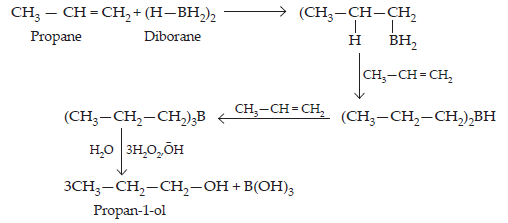
Ans. The addition of diborane to alkenes to form trialkyl boranes followed by their oxidation with alkaline hydrogen peroxide to form alcohols is called hydroboration-oxidation.
For example, propan-1-ol is produced by the hydroboration-oxidation reaction of propene. In this reaction, propene reacts with diborane (BH3)2 to form trialkyl borane as an addition product. This addition product is oxidized to alcohol by hydrogen peroxide in the presence of aqueous sodium hydroxide.
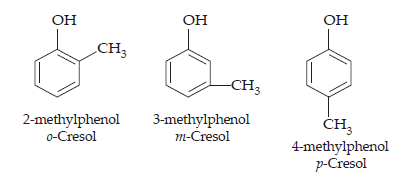
7. Give the structures and IUPAC names of monohydric phenols of molecular formula, C7H8O.
Ans. The three isomers are:
8. While separating a mixture of ortho and para nitrophenols by steam distillation, name the isomer which will be steam volatile. Give reason.
Ans. Ortho-nitrophenol is steam volatile. The reason is chelation due to intramolecule H-bonding. So, it can be separated by steam distillation from p-nitrophenol. Para-nitrophenol is not steam volatile due to presence of intermolecular H-bonding.

9. Give the equations of the reaction for the preparation of phenol from cumene.
Ans. To prepare phenol, cumene is first oxidised in the presence of air to cumene hydroperoxide.

The cumene hydroperoxide thus formed, is treated with dilute acid to prepare phenol and acetone as by-products.

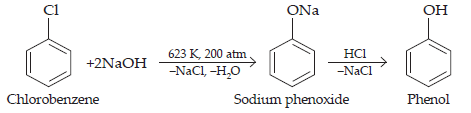
10. Write chemical reaction for the preparation of phenol from chlorobenzene.
Ans. Chlorobenzene is fused with NaOH (at 623 K and 320 atm pressure) to produce sodium phenoxide, which gives phenol on acidification.
11. Write the mechanism of hydration of ethene to yield ethanol.
Ans. The mechanism of hydration of ethene to form ethanol involves three steps:
Step 1: Protonation of ethene to form carbocation by electrophilic attack of H3O+:
$$\text{H}_{2}\text{O}+\text{H}^{+}\xrightarrow{}\text{H}_{3}\text{O}^{\normalsize+}$$
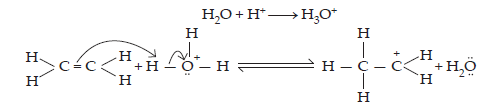
Step 2: Nucleophilic attack of water on carbocation formed:
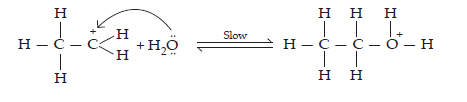
Step 3: Deprotonation to form corresponding alcohols:
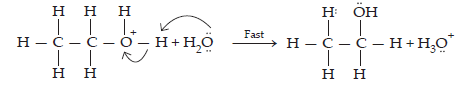
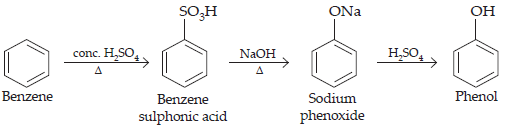
12. You are given benzene, conc. H2SO4 and NaOH. Write the equations for the preparation of phenol using these reagents.
Ans.
13. Show how will you synthesise:
(i) 1-phenylethanol from a suitable alkene.
(ii) Cyclohexylmethanol using an alkyl halide by an SN2 reaction.
(iii) Pentan-1-ol using a suitable alkyl halide?
Ans. (i) 1-phenylethanol can be prepared from ethenylbenzene by addition of H2O in the presence of dil. H2SO4.
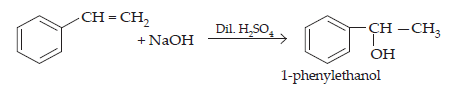
(ii) Hydrolysis of cyclohexylmethyl bromide by aqueous NaOH gives cyclohexylmethanol.
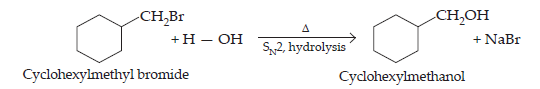
(iii) Hydrolysis of 1-bromopentane by aqueous NaOH gives pentan-1-ol.
$$\underset{\text{1-bromopentane}}{\text{CH}_{3}\text{CH}_{2}\text{CH}_{2}\text{CH}_{2}\text{CH}_{2}\text{Br}}\xrightarrow[\text{S}_{N}^{2}\text{Hydrolysis}]{\Delta}\underset{\text{Pentan-1-ol}}{\text{CH}_{3}\text{CH}_{2}\text{CH}_{2}\text{CH}_{2}\text{CH}_{2}-\text{OH + NaBr}}$$
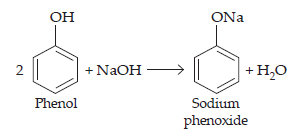
14. Give two reactions that show the acidic nature of phenol. Compare acidity of phenol with that of ethanol.
Ans. The acidic nature of phenol can be represented by the following two reactions:
(a) Reaction with sodium: Phenol reacts with sodium to give sodium phenoxide, liberating H2 gas.

(b) Reaction with NaOH: Phenol reacts with NaOH to form sodium phenoxide and water as a by- product.
Phenol is more acidic than ethanol. This is due to the reason that after the loss of a proton phenol form phenoxide ion which is stabilized by resonance, while ethoxide ion formed after the loss of a proton from ethanol, does not.
The resonating structures of phenoxide ion are shown as below:
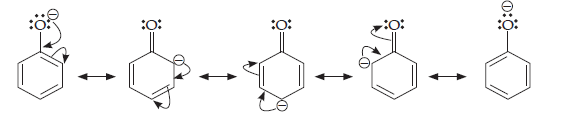
The lone pair of electrons on oxygen delocalizes into the benzene (mesomeric effect) which reduces the electron density in the O—H bond. The O—H bonds are weaker and therefore breaks easily whereas in ethanol the electron releasing inductive effect of the alkyl group increases the electron density on the O—H bond. This strengthens the bond so the bond does break not break easily. Therefore making ethanol less acidic than phenol.
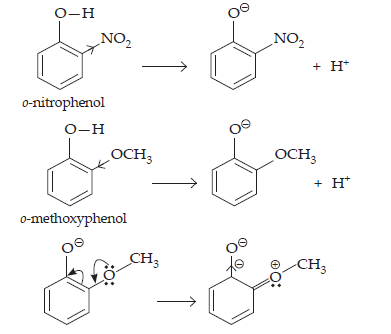
15. Explain why is ortho-nitrophenol more acidic than ortho-methoxyphenol?
Ans.
The nitro group is an electron withdrawing group. The presence of the group in the ortho-position decreases the electron density in the O–H bond. As a result, it is easier to lose a proton. Also, the o-nitrophenoxide ion formed of the loss of proton is stabilised by resonance. Hence, ortho, nitrophenol is a stronger acid.
On the other hand, methoxy group is an electron releasing group. Thus, it increases the electron density in the O–H bond and hence, the proton can not be given out easily.
For this reason, ortho-nitrophenol is more acidic than ortho-methoxyphenol.
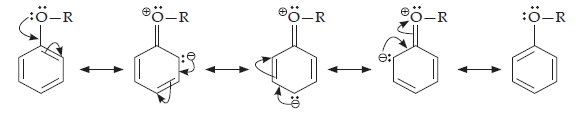
16. Explain how does the – OH group attached to a carbon of benzene ring activate it towards electrophilic substitution?
Ans. Phenol may be regarded as a resonance hybrid of structures I-V, shown below:
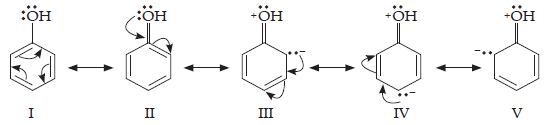
As a result of +R effect of the –OH group, the electron density in the benzene ring increases thereby facilitating the attack of an electrophile. In other words, presence of –OH group, activates the benzene ring towards electrophilic substitution reactions. Further, since the electron density is relatively higher at the two o-and one p-position, therefore electrophilic substitution occurs mainly at o-and p-positions.
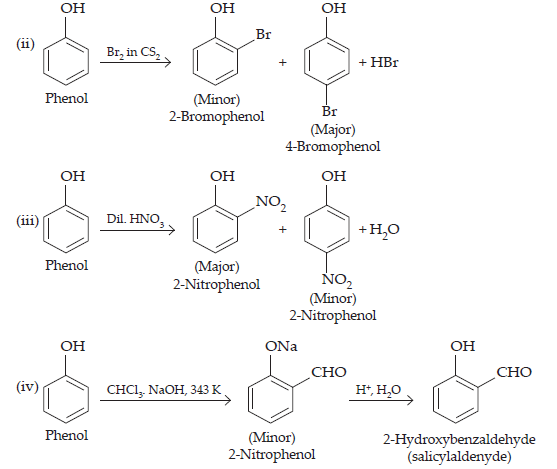
17. Give equations of the following reactions:
(i) Oxidation of propan-1-ol with alkaline KMnO4 solution.
(ii) Bromine in CS2 with phenol.
(iii) Dilute HNO3 acid with phenol
(iv) Treating phenol with chloroform in presence of aqueous NaOH.
Ans.
$$\underset{\text{Propan-1-ol}}{\text{CH}_{3}\text{CH}_{2}\text{CH}_{2}\text{OH} + 2[\text{O}]}\xrightarrow[\text{Oxidation}]{\text{Alk.kMnO}_{4}}\text{CH}_{3}\text{CH}_{2}\text{COOH + H}_{2}\text{O}$$
18. Explain the following with an example:
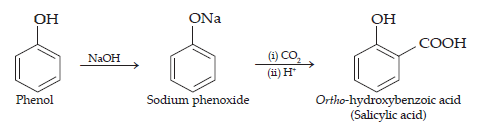
(i) Kolbe’s reaction.
(ii) Reimer – Tiemann reaction.
(iii) Williamson ether synthesis.
(iv) Unsymmetrical ether.
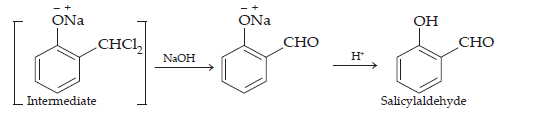
Ans. (i) Kolbe’s reaction: When phenol is treated with sodium hydroxide, sodium phenoxide is produced. This sodium phenoxide when treated with carbon dioxide, followed by acidification, undergoes electrophilic substitution to give ortho-hydroxybenzoic acid (salicylic acid) acid as the main product. This reaction is known as Kolbe’s reaction.
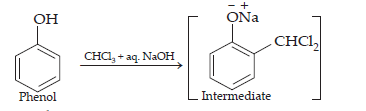
This reaction is known as the Reimer-Tiemann reaction.
The intermediate is hydrolysed in the presence of alkalis to produce salicylaldehyde.
(iii) Williamson ether synthesis: Williamson ether synthesis is a laboratory method to prepare symmetrical and unsymmetrical ethers by allowing alkyl halides to react with sodium alkoxides.
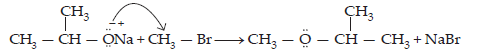
If the alkyl halide is secondary or tertiary, then elimination competes over substitution.
(iv) Unsymmetrical ether: An ether in which alkyl or aryl groups attached to the oxygen atom are different, is known as unsymmtrical ether. Example: Ethyl methyl ether (C2H5OCH3), methyl phenyl ether (CH3—O—C6H5).
19. Write the mechanism of acid dehydration of ethanol to yield ethene.
Ans. The mechanism of dehydration of alcohols to form alkenes occur by the following three steps:
(a) Formation of protonated alcohol:
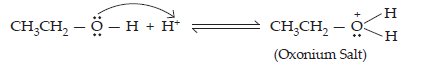
(b) Formation of carbocation:
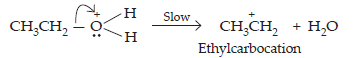
(c) Elimination of a proton to form ethene:
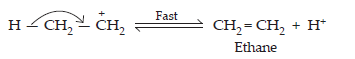
20. How are the following conversions carried out?
(i) Propene to Propan-2-ol
(ii) Benzyl chloride to Benzyl alcohol
(iii) Ethyl magnesium chloride to Propan-1-ol
(iv) Methyl magnesium bromide to 2-Methylpropan-2-ol
Ans. (i) When propene is allowed to react with water in the presence of an acid as a catalyst, then propan-2-ol is obtained. The process of conversion of propene to propan-2-ol is also called as acid hydration.
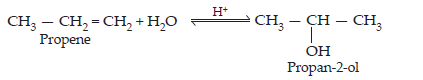
(ii) If benzyl chloride is treated with NaOH (followed by acidification) then benzyl alcohol is produced.
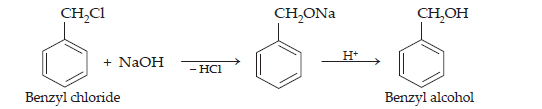
(iii) When ethyl magnesium chloride is treated with methanal in the presence of dry ether , an addition product is obtained which gives propan-1-ol on hydrolysis.
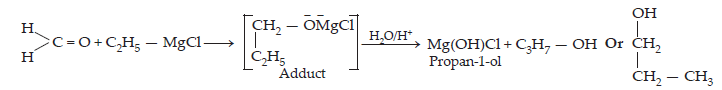
(iv) When methyl magnesium bromide is treated with propane in the presence of dry ether , an adduct is obtained which gives 2-methylpropan-2-ol on hydrolysis.
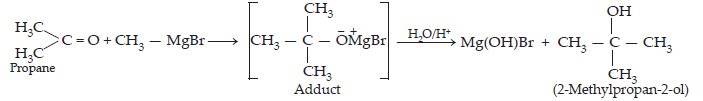
21. Name the reagents used in the following reactions:
(i) Oxidation of a primary alcohol to carboxylic acid.
(ii) Oxidation of a primary alcohol to aldehyde.
(iii) Bromination of phenol to 2,4,6-tribromophenol
(iv) Benzyl alcohol to benzoic acid.
(v) Dehydration of propan-2-oI to propene.
(vi) Butan-2-one to butan-2-ol .
Ans. (i) Acidified potassium dichromate or neutral/acidic/alkaline potassium permanganate.
(ii) Pyridinium chlorochromate (PCC), (C5H5NH) + ClCrO3– in CH2Cl2 or Pyridinium dichromate (PDC), [(C5H5NH)2]2+ Cr2O72– in CH2Cl2
(iii) Aqueous bromine, i.e., Br2/H2O.
(iv) Acidified or alkaline potassium permanganate.
(v) 85% H2SO4 at 440 K.
(vi) Ni/H2 or NaBH4 or LiAlH4.
22. Give reason for the higher boiling point of ethanol in comparison to methoxymethane.
Ans. Hydrogen bonding is present between the ethanol molecules which is absent in molecules of methoxy methane. So, a large amount of energy is required to break these strong bonds in ethanol.
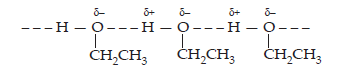
23. Give IUPAC names of the following ethers:
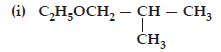
(ii) CH3OCH2CH2Cl
(iii) O2N — C6H4 —OCH3
(iv) CH3CH2CH2OCH3
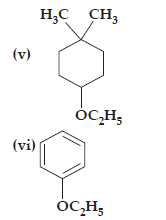
Ans. (i) 1-Ethoxy-2-methylpropane
(ii) 2-Chlorlo-l-methoxyethane
(iii) 4-Nitroanisole
(iv) 1-Methoxypropane
(v) 1-Ethoxy-4-4 – dimethyl cyclohexane
(vi) Ethoxybenzene
24. Write the names of the reagents and equations for the preparation of the following ethers by Williamson’s synthesis :
(i) 1-Propoxypropane
(ii) Ethoxybenzene
(iii) 2-Methoxy-2-methylpropane
(iv) Methoxyethane.
Ans. (i)
$$\underset{\text{1-Bromopropane}}{\text{CH}_{3}\text{CH}_{2}\text{CH}_{2}\text{CH}_{2}\text{Br} } + \underset{\text{Sod. propoxide}}{\text{CH}_{3}\text{CH}_{2}\text{CH}_{2}\text{ONa}}\xrightarrow{}\underset{\text{1-Propoxypropane}}{\text{CH}_{3}\text{CH}_{2}\text{CH}_{2}\text{O CH}_{2}\text{CH}_{2}\text{CH}_{3} + \text{NaBr}}$$
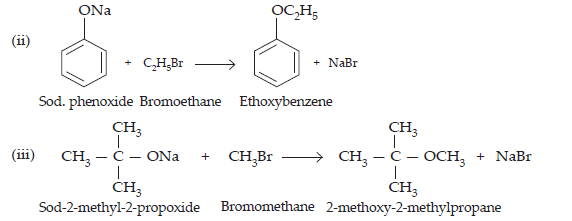
$$\text{(iv)}\space\underset{\text{Sod. ethoxide}}{\text{C}_{2}\text{H}_{5}}+\underset{\text{Bromomethane}}{\text{CH}_{3}\text{Br}}\xrightarrow{}\underset{\text{Methoxyethane}}{\text{C}_{2}\text{H}_{5}-\text{O}-\text{CH}_{3} + \text{NaBr}}$$
25. Illustrate with examples the limitations of Willamson synthesis for the preparation of certain types of ethers.
Ans. Williamson’s synthesis is a versatile method for the synthesis of both symmetrical and unsymmetrical ethers. However, for the synthesis of unsymmetrical ethers, a proper choice of reactants is necessary. Since Williamson’s synthesis occurs by SN2 mechanism and primary alkyl halides are most reactive in SN2 reaction, therefore, best yields of unsymmetrical ethers are obtained when the alkyl halides are primary and the alkoxide may be primary, secondary or tertiary. For example, tert-butylethyl ether is prepared by treating ethyl bromide with sodium tert-butoxide.
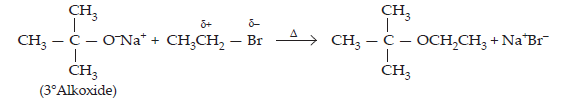
The above ether cannot be prepared by treating sodium ethoxide with tert-butyl chloride or bromide since under these condition an alkene, i.e., isobutylene is the main product.
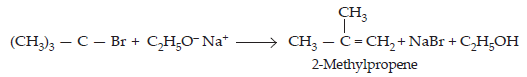
Aryl and vinyl halides cannot be used as substrates because they are less reactive in nucleophilic substitution.
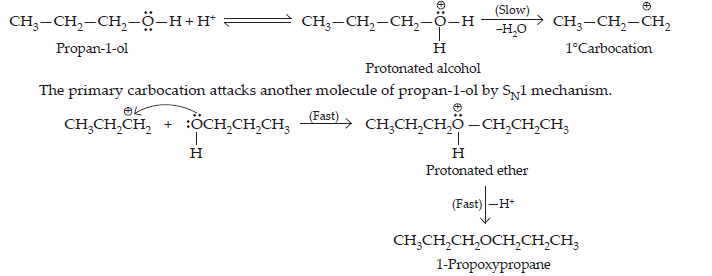
26. How is 1-propoxypropane synthesised from propan-1-ol? Write mechanism of the reaction.
Ans. Any one of the following methods can be used for the synthesis of 1-propoxypropane from propan-1-ol.
(i) By Williamson’s synthesis
The halogen derivative such as bromoderivative and sodium salt of the alcohol take part in the Williamson’s synthesis.
$$\underset{\text{Propan-1-ol}}{\text{(a)\space}3\text{CH}_{3}-\text{CH}_{2}-\text{CH}_{2}-\text{OH} + \text{PBr}_{3}}\xrightarrow{}\underset{\text{1-Bromopropane}}{3\text{CH}_{3}-\text{CH}_{2}-\text{CH}_{2}-\text{Br} + \text{H}_{3}\text{PO}_{3}}\\\underset{\text{Propan-1-ol}}{\text{(b)\space}\text{CH}_{3}-\text{CH}_{2}-\text{CH}_{2}-\text{OH} + \text{Na}}\xrightarrow{}\underset{\text{Sodium propoxide}}{\text{CH}_{3}-\text{CH}_{2}-\text{CH}_{2}-\text{O}^{\normalsize-}\text{Na}^{\normalsize+}+1/2\text{H}_{2}}$$
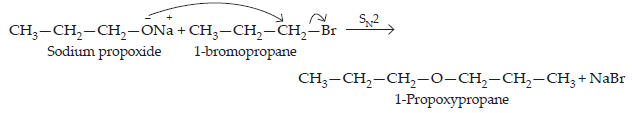
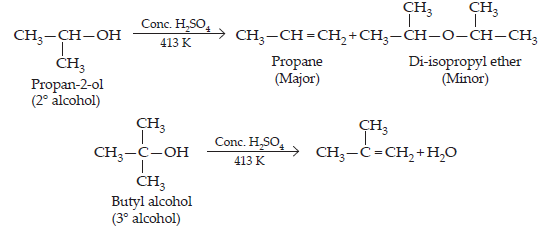
27. Preparation of ethers by acid dehydration of secondary or tertiary alcohols is not a suitable method. Give reason.
Ans. Ethers are formed as a result of acid dehydration by SN2 mechanism (from primary alcohols). If secondary or tertiary alcohols are used, due to steric hinderance, alkenes are formed and not ethers.
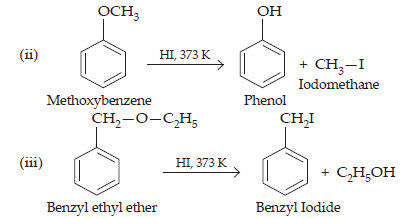
28. Write the equation of the reaction of hydrogen iodide with (i) 1-propoxypropane (ii) methoxybenzene, and (iii) benzyl ethyl ether.
$$\textbf{Ans.\space}\text{(i)}\space\text{CH}_{3}\text{CH}_{2}\text{CH}_{2}\text{O}\text{CH}_{2}\text{CH}_{2}\text{CH}_{3}\xrightarrow[\text{373 K}]{\text{HI}}\underset{\text{Propan-1-ol}}{\text{CH}_{3}\text{CH}_{2}\text{CH}_{2}-\text{OH} + }\underset{\text{Iodopropane}}{\text{CH}_{3}\text{CH}_{2}\text{CH}_{2}\text{I}}$$
29. Explain the fact that in alkyl aryl ethers:
(i) Alkoxy group activates the benzene ring towards electrophilic substitution.
(ii) It directs the incoming substituents towards ortho and para positions in benzene ring.
Ans. (i) In aryl alkyl ethers, +R effect of the alkoxy group (–OR) increases the electron density in the benzene ring thereby activating the benzene ring towards electrophilic substitution reactions.
As the ortho and para positions in the ring become points of high electron density, the electrophiles prefer to attack these positions.
(ii) The alkoxy group directs the incoming group which is an electrophile towards the ortho and para positions in the ring. As a result, a mixture of isomeric products is formed.
30. Write the mechanism of the reaction of HI with methoxymethane.
Ans. The mechanism of the reaction of HI with methoxymethane involves the following steps:
Step 1: Protonation of methoxymethane:
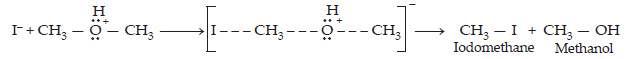
Step 3: When HI is in excess and the reaction is carried out at a high temperature, the methanol formed in the second step reacts with another HI molecule and gets converted to methyl iodide.
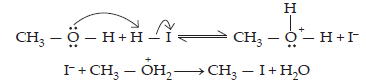
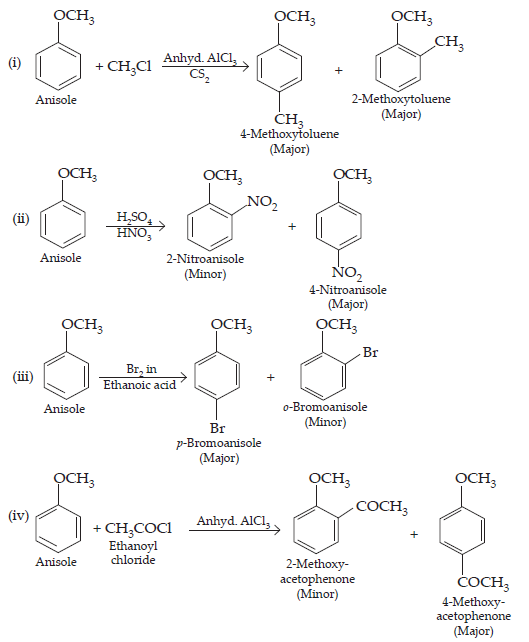
31. Write equations of the following reactions:
(i) Friedel-Crafts reaction -alkylation of anisole
(ii) Nitration of anisole.
(iii) Bromination of anisole in ethanoic acid medium
(iv) Friedel-Craft’s acetylation of anisole.
Ans.
32. Show how will you synthesise the following alcohols from appropriate alkenes:
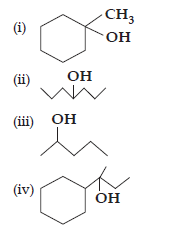
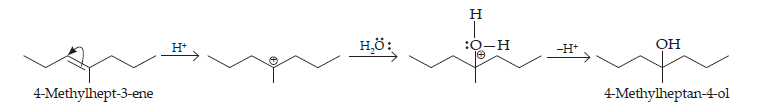
Ans. All the given alcohols can be synthesised by the acid-catalysed hydration of alkenes in the acidic medium. The addition follows Markovnikov’s rule.
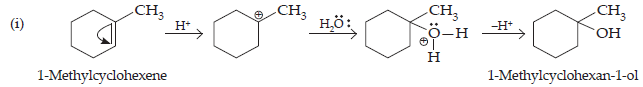
(ii) 4-Methylpent-3-ene upon hydration in the acidic medium will give the desired alcohol.
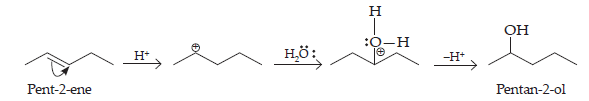
(iii) Pent-2-ene gives the desired alcohol upon hydration in the presence of acid.
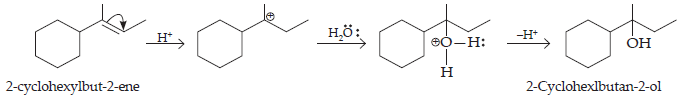
(iv) The cyclic alkene used in this reaction is 2-cyclohexylbut-2-ene.
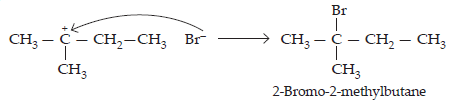
33. When 3-methylbutant 2-ol is treated with HBr, the following reaction takes place:
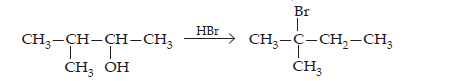
Give a mechanism for this reaction.
(Hint: The secondary carbocation formed in step II rearranges to a more stable tertiary carbocation by a hydride ion shift from 3rd carbon atom.)
Ans. The mechanism of the given reaction involves the following steps:
Step 1: Protonation
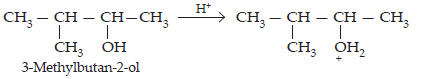
Step 2: Formation of 2° carbocation by the elimination of water molecule
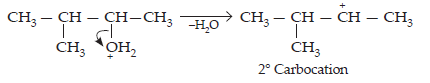
Step 3: Re-arrangement by the hydride-ion shift
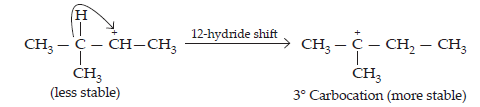
Step 4: Nucleophilic attack